The Challenge: Moving Beyond ‘Blind’ Immunosuppression

For kidney transplant recipients, the gift of a new organ comes with a lifelong trade-off: the constant use of immunosuppressive drugs. While essential to prevent the body from rejecting the graft, these medications at high doses can lead to severe side effects including chronic infections, cardiovascular issues, and kidney toxicity.
Physicians therefore often face a difficult choice: maintain standard drug levels and risk toxicity, or reduce the dose and risk a “silent”, subclinical rejection (SCR). SCR occurs when the immune system quietly infiltrates the kidney, causing microscopic inflammation and cellular damage that remains invisible to standard blood tests. Because this damage does not impact immediate kidney function, it can go undetected for years, only becoming apparent once permanent scarring has formed and it is often too late to save the organ. In the past, SCR could only be detected through invasive surveillance biopsies, which are painful, carry risks of bleeding, and are not performed in all hospitals. There is currently no harmonised protocol to guide physicians when to reduce immunosuppressive therapy or how to safely monitor patients afterwards. To improve the lives of these patients, a technique is required that can detect the beginnings of SCR without the painful and risky biopsies.
A Breakthrough in Predicting Graft Stability
The AGORA (Algorithm of Graft Outcome in Renal Allotransplantation) project, an ambitious multinational consortium funded by ERA PerMed (JTC2021), was built upon a vital scientific foundation. Before the consortium was launched, a landmark study published in Kidney International provided the proof-of-concept required to move forward.
This foundational research searched for biomarkers in the blood that could act as early warning signs of SCR, and hence serve as a non-invasive “liquid biopsy” that could replace the risky and painful kidney biopsy procedure. A key scientific breakthrough of the project was the validation of combined biomarkers. The team confirmed that combining clinical data that forms the Kidney Transplant Failure Score (KTFS) with a specific gene expression signature (SCR-S) provides superior predictive power compared to using clinical data alone.
By tracking a drop in this gene signature, the AGORA algorithm can spot when the immune system begins preparing for a “silent” attack. The precision of the SCR-S score is exceptional, achieving a Negative Predictive Value (NPV) of 0.977, meaning it can rule out subclinical rejection with near-total certainty. This provides clinicians with the confidence needed to identify low-risk patients who can safely benefit from reduced medication.
AGORA Study Design
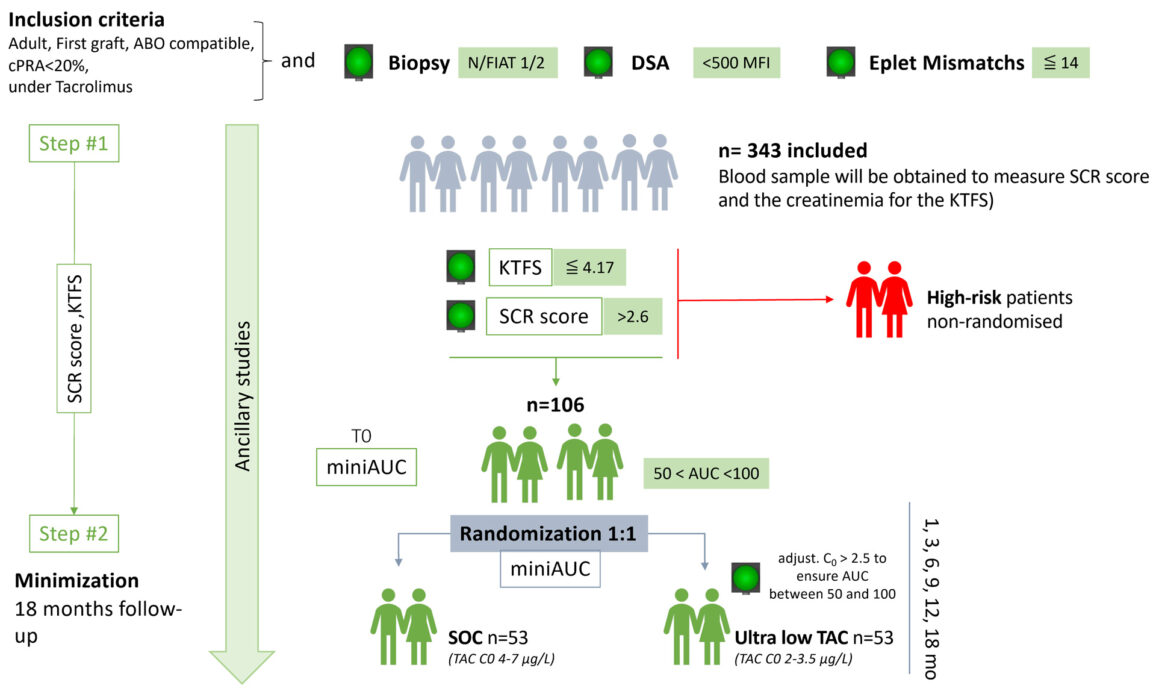
The stability and reliability of this approach were further established through a retrospective analysis of over 300 patients from the DIVAT cohort- a large-scale, multicenter French database that tracks long-term outcomes for transplant recipients. Using this “real-world” clinical data as a proving ground, this study validated that the AGORA algorithm effectively stratifies patients into distinct risk categories as early as one year post-transplant. Instead of waiting for detectable kidney damage, the research team identified a biological signature in the blood that shifts before the immune system causes permanent injury.
This level of precision provides an early warning system allowing doctors to identify high-risk patients who need protection before scarring occurs, while safely reducing the amount of medication for low-risk patients to avoid toxic side effects. This personalised strategy helps ensure long-term graft stability and a higher quality of life for transplant patients.
From Discovery to Clinical Reality
The AGORA algorithm is currently being tested within the AGORAC clinical trial framework. This Phase III, multicenter study is currently validating the safe minimisation of immunosuppression in 106 recipients across France, Spain, and Norway. To ensure the AGORA tool is practical for daily life, the project has integrated a “finger-prick” test for at-home monitoring. This innovation empowers patients to monitor their graft health from home, reducing the need for frequent and stressful hospital visits.
Recognising Excellence: The Legion of Honor

The profound impact of this research journey reached the highest level of national recognition in 2025. Prof. Sophie Brouard, the AGORA consortium leader, was appointed Knight of the Legion of Honor (Chevalier de la Légion d’honneur), the highest French order of merit. For Prof. Brouard and her team, this distinction represents more than a personal milestone; it is a validation of a “collective adventure” that has spanned over twenty years, bridging the gap between immunology, clinical innovation, and patient care.
The Evolution of Stratified Medicine: BATMAN
The philosophy established in the successful AGORA project has been applied to a new consortium project funded under EP PerMed JTC2024, BATMAN. While AGORA focused on the “silent” signals of kidney transplant rejection, the BATMAN consortium is applying this expertise to Sjögren’s disease (SjD) – a systemic autoimmune disorder where the immune system attacks moisture-producing glands, significantly increasing the risk of B-cell lymphoma. The team now aims to validate the BOB1 protein as a pivotal biomarker and develop a novel inhibitor for personalised treatment to stop the transition to lymphoma before it begins.
The Power of Patient Engagement and Multinational Collaboration
The AGORA project further incorporated a sociological work package that empowered patients to share their perspectives. Through interviews and focus groups, the team ensured the AGORA algorithm is ethically grounded, user-friendly, and widely accepted by both patients and medical staff. This patient-centered approach is designed to improve treatment adherence and ensure long-term continuity of care.
The sociological work package and clinical study were implemented across all countries of the multinational collaboration – France, Spain, and Norway, allowing for a larger, more diverse patient pool, as well as the harmonisation of laboratory protocols across borders. Coordinated by Sophie Brouard (CNRS, CR2TI, 1064 INSERM Unit, Centre Hospitalier Universitaire de Nantes, France), this initiative brought together leading international experts including Magali Giral (Clinical coordinator, Centre Hospitalier Universitaire de Nantes, France), Oriol Bestard (Partner 1, Fundació Hospital Universitari Vall d’Hebron, Spain), Anders Åsberg (Partner 2, Oslo University Hospital, Norway) and Nicolas Degauque (Partner 3, Université de Nantes, France). By integrating patient perspectives into rigorous science, the consortium has moved the AGORA algorithm from the laboratory to a clinical reality, with the promise of safeguarding the health and stability of transplant patients.
This work is vital because it finally allows us to treat transplant patients as individuals rather than clinical averages. By using precision biomarkers to safely guide treatment reduction, we are not just protecting the graft; we are significantly improving the daily quality of life for the patients.
Dr. Sophie Brouard, Project Coordinator/ Pr. Magali Giral, Clinical coordinator.
The AGORA project was supported by
- French National Research Agency (ANR)
- The Research Council of Norway (RCN)
- Carlos III Health Institute (ISCIII)
under the frame of ERA PerMed JTC2021.
The BATMAN project is supported by
- French National Research Agency (ANR)
- Federal Ministry of Health (BMG)
- Federal Ministry of Research, Technology and Space (BMFTR)
- Health Research Board (HRB)
- Ministero dell’università e della ricerca (MUR)
- The Netherlands Organisation for Health Research and Development (ZonMw)
under the frame of EP PerMed JTC2024.

