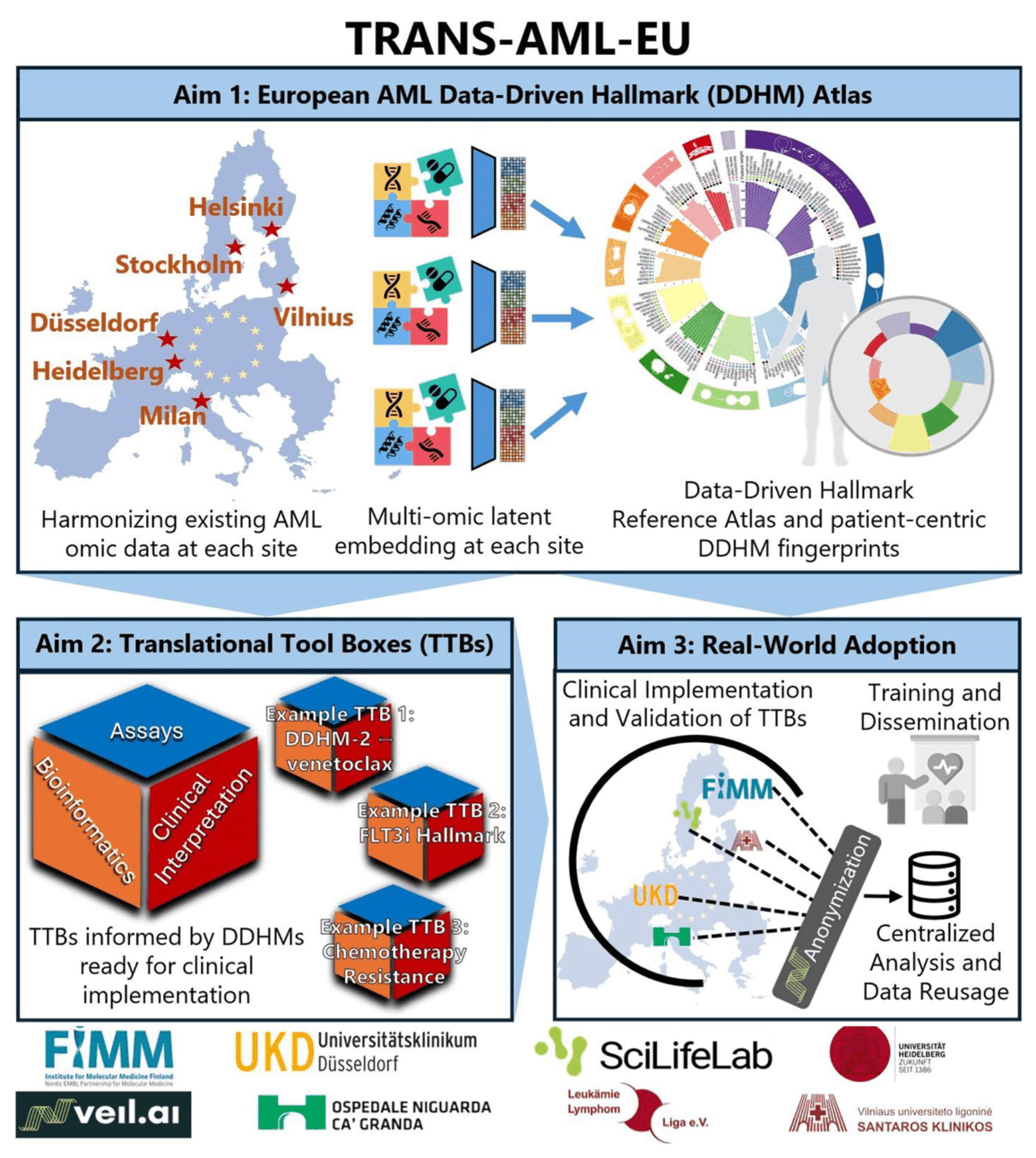
TRANS-AML-EU aims to transform how therapies are selected for patients with acute myeloid leukemia (AML). Rather than relying on single mutations to categorise patients into disease subgroups, the project combines genomics, epigenomics, transcriptomics, proteomics and ex vivo drug-response profiling to derive latent disease programs, termed Data-Driven Hallmarks (DDHMs). These DDHMs are derived by combining data from more than 1,000 AML cases using integrative, unsupervised modelling and capture how leukemia cells behave and respond to drugs. Each patient is assigned a vector of DDHMs, enabling multi-axis precision medicine beyond the “one mutation, one drug” paradigm.
To translate these discoveries into practice, TRANS-AML-EU develops Translational Tool Boxes (TTBs) that distil actionable hallmarks into (i) minimal, clinic-ready biomarker panels, (ii) validated prediction or stratification algorithms, and (iii) standard operating procedures for laboratory and bioinformatic implementation. TTBs are designed for rapid turnaround (within a few days) on standard hospital platforms, allowing decentralized use across diverse health-care systems.
Retrospective outcome analyses and a prospective, multicenter “clinical trial in a dish” will benchmark TTB predictions against clinical and ex vivo responses. Patient and family perspectives are integrated through active involvement of the Leukämie Lymphom Liga. Ultimately, TRANS-AML-EU seeks to establish a transferable framework for hallmark-based pharmacogenomics that can be extended beyond AML to other cancers and diseases.
The consortium is still under contract negotiations and changes may occur.

