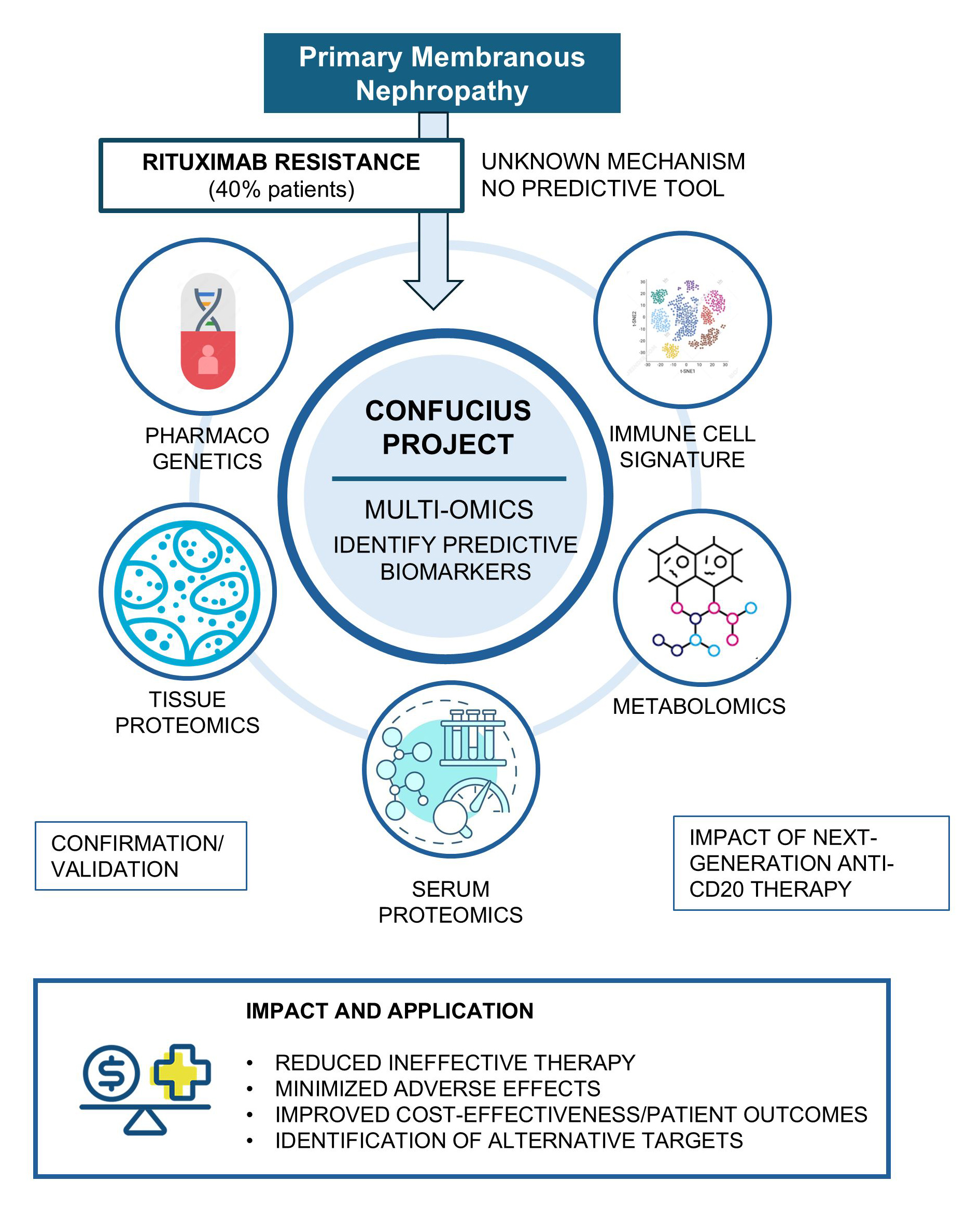
Primary membranous nephropathy (MN) is a leading cause of nephrotic syndrome in adults resulting from an abnormal immune response in which autoantibodies attack the renal filtration barrier. Rituximab (RTX), an anti-CD20 antibody that depletes the immune cells responsible for producing these autoantibodies, is increasingly used as a first-line treatment, achieving remission in many patients. However, in around 4 out of 10 patients the treatment does not work, exposing individuals to side effects without any real benefit. Currently, we do not know why some patients fail to respond. The mechanisms underlying RTX resistance remain unclear and no tools are available to predict treatment response.
The CONFUCIUS project aims to establish a personalised medicine framework for MN through a multi-omics approach. Genetic variants, serum and kidney proteomic profiles, and serum metabolomic profiles will be analysed in a well-characterized retrospective patient cohort to identify biomarkers capable of predicting whether a patient will respond to rituximab, thereby enabling more accurate treatment selection. Immune cells will also be examined at single-cell resolution to determine cellular features associated with resistance.
Findings will be validated in an independent cohort. The project will also assess whether obinutuzumab, a next-generation anti-CD20 antibody, can provide clinical benefit in RTX-nonresponsive patients. If obinutuzumab also fails, the multi-omics strategy will be reapplied to clarify the underlying mechanisms and identify new potential therapeutic targets. Finally, the economic and social impact of a treatment strategy guided by pharmacogenomics and multi-omics technologies will be evaluated to determine its cost-effectiveness and potential to improve patient care.

