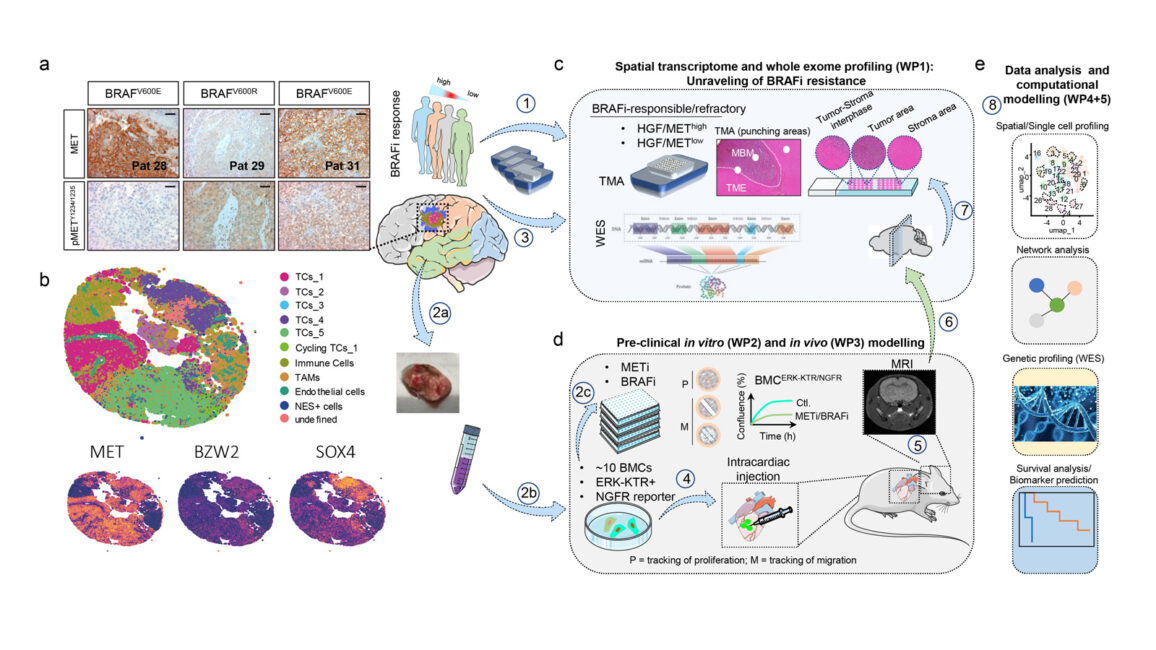
Description of figure “Project workflow”
a. Immunohistochemistry of BRAF-mutant MBM showing expression (MET) and activation (pMETY1234/1235) of MET. b. Spatial mapping of MET+ cellular subsets (top) and expression levels of MET, BZW2 and SOX4 (bottom) c. (1) Archived (FFPE) BRAFi-responsive/naïve and refractory MBM will be subjected to TMA preparation and subsequent spatial transcriptomics analyses. d. (2a) MBM-derived cell lines (BMCs) have been established from intraoperative tumors. (2b) Lentiviral transduction of BMCs with ERK-KRT and NGFR reporters. (2c) Established reporter cells will be seeded on 96-well plates and treated with BRAFi, METi and combinations; the efficacy of inhibitors will be assessed by live cell-imaging-based tracking of cell migration (M) and proliferation (P). Imaging will determine the fluorescence intensity of reporters and cellular confluence. (3) Tumor DNA will be used for whole-exome sequencing (WES). (4) Brain-penetrable METi, BRAFi and METi/BRAFi treatment following intracardiac injection of BMC reporter cells; (5) Brain tumour growth will be monitored via MRI and (6),(7) tumour bearing brains will be resected and subjected to single-cell resolved spatial transcriptomics (scST). e. (8) Data analysis and computational modelling will comprise single-cell expression profiling, network analysis, genetic profiling and survival analyses and biomarker identification.
Background
First-line therapies that inhibit the overactivated BRAF kinase (BRAFi) initially show high effectiveness but fail to control melanoma brain metastases (MBM) durably due to acquired or intrinsic resistance mechanisms. Therapy-resistant tumour cells lead to progressive intracranial disease in 40-60% of patients and a poor prognosis. The consortium already discovered activation of the tyrosine kinase receptor MET in MBM, possibly mediating resistance to therapy.
Hypothesis
Intracranial resistance is genetically and spatially controlled by activation of MET-driven molecular programs determining the therapeutic response of MBM to BRAFi. The consortium proposes that brain-penetrable ATP-competitive small molecule MET inhibitors (METi) in combination with BRAFi may represent a promising strategy to combat tumour recurrence in the brain and improve the survival of patients with BRAFi-refractory melanoma.
Aims
The pharmacogenomic study aims on
- Identification of genetic aberrations, spatial tumour-stromal interaction mechanisms and regulators that promote MET activation and thus resistance to BRAFi in MBM.
- Development of cellular reporters for live-cell imaging-based tracking of MAPK-activation downstream of MET receptor and for in vitro testing of highly effective tumour cell-eliminating METi/BRAFi combinations.
- Evaluation of the effect of METi/BRAFi on the cellular composition of established brain tumours in preclinical in vivo models in response to BRAFi/METi.
- Determination of pharmacogenomic biomarkers and molecular signatures for predicting therapy response and resistance. The consoritum’s vision is to combat therapy-resistance in patients with MBM.
- Explore our findings’ translation path, supported by industry and healthcare stakeholder groups.
The project in a nutshell
The project´s main aim and the consoritum’s vision is to combat therapy-resistance in patients with melanoma brain metastases through spatial transcriptomic and genetic profiling and pre-clinical and in-silico models.

